PREMIUM PAIN MANAGEMENT PRODUCTS
LCCS MEDICAL PRODUCTS
WHAT MAKES US A GREAT PARTNER?
We are committed to being the most trusted supply partner.
We offer nationwide distribution from our U.S. headquarters in Arizona.
We focus on delivering superior customer service to our clients.

Our goal is to help improve patients’ lives by offering high quality medical products.

History of LCCS Medical Inc.
LCCS Medical Inc. is a privately held company markets and distributes premium quality specialty medical devices for pain management and regional anesthesia since 2019. Every device is designed precisely for our physician partners to aid in the comfort of patients while also bettering surgical outcomes
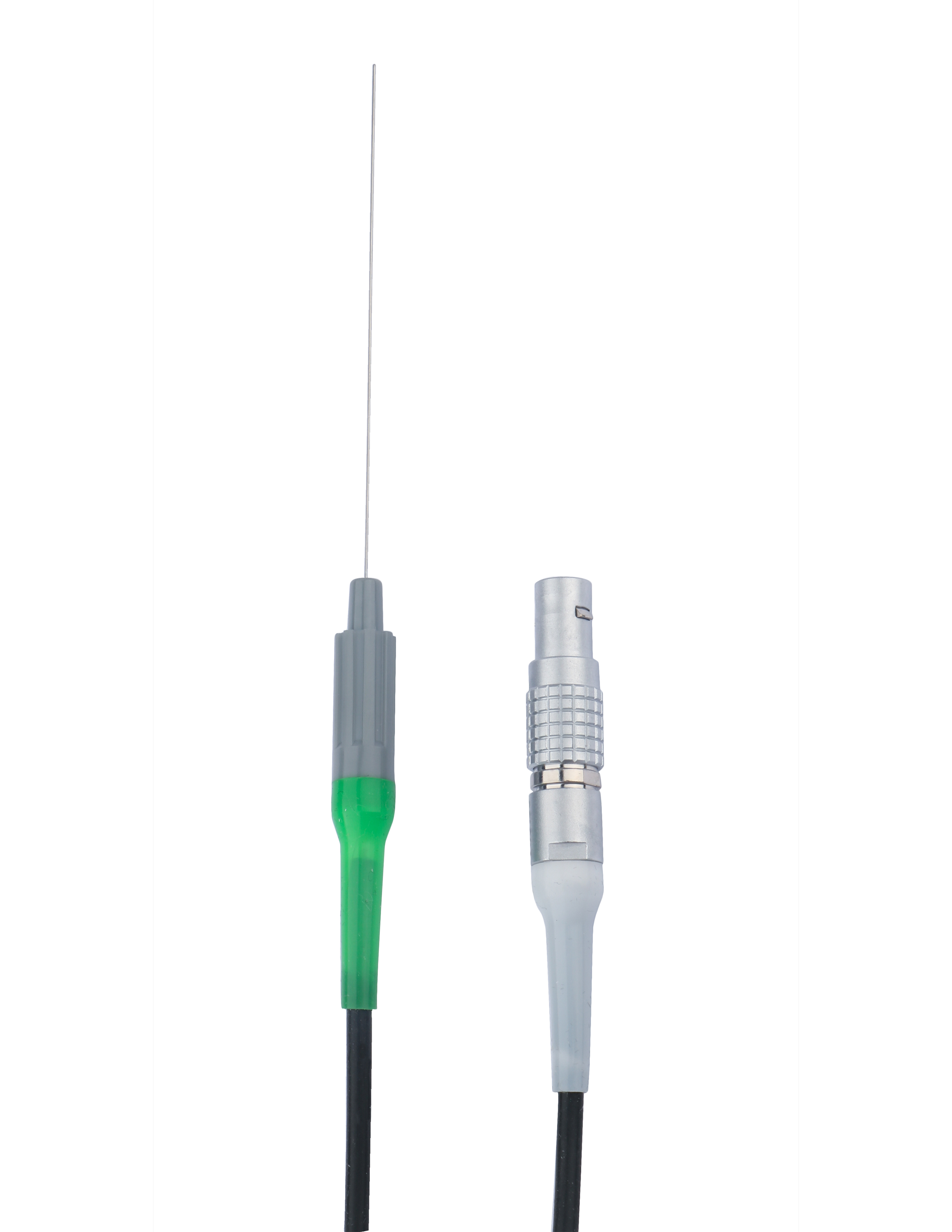
Our RF Cannulas, RF Electrodes, and Grounding Pads, along with Spinal Needles and Epidural Needles are U.S. FDA approved. Additionally, the RF Cannula and RF Electrodes are CE marked. Our U.S. headquarters distributes these physicians’ preferred devices to customers nationwide.
Contact us
The focus of our team is delivering superior service to our clients. For more information on our range of high quality products, please email Info@LCCSmedical.com or call our team at 844-743-6449.

About
LCCS Medical Inc. started developing RF pain management and regional anesthesia products in 2009. We started selling these products to the U.S. market in 2018. Since 2018, we have had zero FDA incidents.